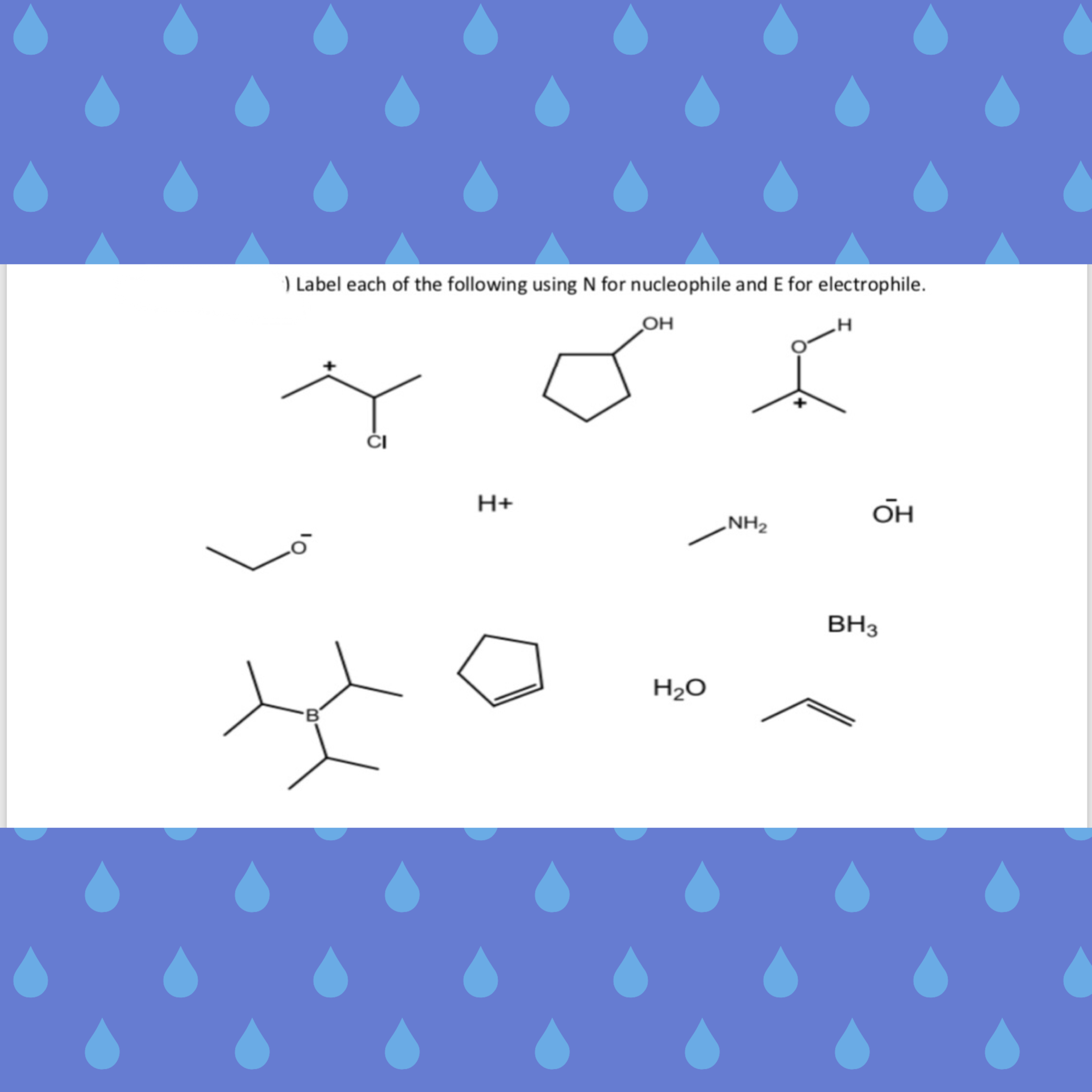
Here, R is an alkyl group (we can use methyl, ethyl, or any alkyl group here), X is a halogen, such as C 2 H 5 - Mg - ClĪs the bond breaks, a partial charge on this compound forms in the following way: This means that compounds in which metal is bonded with C-atom are organometallic compounds. The compounds having organic and metallic compounds joined together are organometallic compounds. Nucleophiles that become neutral because of the lone pair of electrons are neutral. The examples of these nucleophiles are Cl ⊝, Br ⊝, CH - 3, OH -, NH 2 -, O 2 2-, C 2 H 5 O. The nucleophiles that are negatively charged are electron-rich species, and they attack at the electron-deficient center. They attack at the electron-deficient center because of the excess of electrons. These species have lone pairs of electrons. So, polar functions are electrophiles and nucleophiles. Here, C is electron-deficient, so it is an electrophile, while O is electron-rich, so it is a nucleophile. Other examples of incomplete octet electrophiles are Cl ⊕, CH 3 ⊕, CH įor example, Oxygen captures electrons from the C-atom, and forms partial charge, as shown below: That’s why the order becomes BI 3 > BBr 3 > BCl 3 > BF 3. Also, the π-bond between B and F is stronger, which makes it unstable, while BI 3 is the strongest because the size of I is bigger. In all these compounds, back bonding occurs but what happens here is that the size of F is smaller, as it gets additional electrons from Boron, it gives the electron back to Boron through back bonding. Let’s find the order of the strength of an electrophile among BF 3, BCl 3, BBr 3, BI 3. In this structure, what happens is, Boron has empty p-orbital combines with p-orbital of fluorine, where fluorine donates its lone pair to Boron and the bonding formed between them is the back bonding, as you can see in the image below: 
Through this structure, we can see that BF 3 is an electron-deficient species. 
Let’s take examples: BF 3, BCl 3, BBr 3, BI 3, BeCl 2, AlCl 3. However, if NH 4 ⊕, H 3 O ⊕ is in an aqueous medium, i.e., when allowed to dissociate. That’s why all positively charged species aren’t electrophiles. 
So, electrons don’t find any place for them. There are three H + atoms, each combined with 2p-orbitals of N, and the fourth H + atom combines with 2s-orbital of N, to take a lone pair of electrons and fill its octet. It’s because all the orbitals of N are O and are occupied by H-atoms. Positively Charged ElectrophileĮlectrophile examples: H ⊕, Cl ⊕, Br ⊕, CH 3 ⊕Īll positively charged species are not electrophiles. Let’s discuss the three types of electrophiles: 1. These attacking reagents attack at the electron-rich center, i.e., at the substrate. Let’s understand these two terms one-by-one:Įlectron-deficient species are electrophiles. The word ‘phile’ in nucleophile means a nucleus lover. The word ‘phile’ in electrophile means lover, so the word electrophile means electron lover. There are two types of attacking reagents, which are: So, under the reaction mechanism, we are going to study the electrophile and nucleophile. This nucleophile makes changes to form a product P. R is the attacking reagent that can be an electrophile or a nucleophile. Here, S is the substrate or a reactant (a molecule on which the reaction is occurring). Let’s consider a chemical equation, where a reactant transforms into a product. We study general organic chemistry, which talks about all the general things that are backbones of organic chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
